Across all treatment phases, the incidence of treatment-related adverse events of grade 3 or higher was 78.0% in the pembrolizumab–chemotherapy group and 73.0% in the placebo–chemotherapy group, including death in 0.4% (3 patients) and 0.3% (1 patient), respectively. 
After a median follow-up of 15.5 months (range, 2.7 to 25.0), 58 of 784 patients (7.4%) in the pembrolizumab–chemotherapy group and 46 of 390 patients (11.8%) in the placebo–chemotherapy group had disease progression that precluded definitive surgery, had local or distant recurrence or a second primary tumor, or died from any cause (hazard ratio, 0.63 95% CI, 0.43 to 0.93). ResultsĪt the first interim analysis, among the first 602 patients who underwent randomization, the percentage of patients with a pathological complete response was 64.8% (95% confidence interval, 59.9 to 69.5) in the pembrolizumab–chemotherapy group and 51.2% (95% CI, 44.1 to 58.3) in the placebo–chemotherapy group (estimated treatment difference, 13.6 percentage points 95% CI, 5.4 to 21.8 P<0.001). The primary end points were a pathological complete response at the time of definitive surgery and event-free survival in the intention-to-treat population. After definitive surgery, the patients received adjuvant pembrolizumab or placebo every 3 weeks for up to nine cycles. In this phase 3 trial, we randomly assigned (in a 2:1 ratio) patients with previously untreated stage II or stage III triple-negative breast cancer to receive neoadjuvant therapy with four cycles of pembrolizumab (at a dose of 200 mg) every 3 weeks plus paclitaxel and carboplatin (784 patients the pembrolizumab–chemotherapy group) or placebo every 3 weeks plus paclitaxel and carboplatin (390 patients the placebo–chemotherapy group) the two groups then received an additional four cycles of pembrolizumab or placebo, and both groups received doxorubicin–cyclophosphamide or epirubicin–cyclophosphamide. Whether the addition of pembrolizumab to neoadjuvant chemotherapy would significantly increase the percentage of patients with early triple-negative breast cancer who have a pathological complete response (defined as no invasive cancer in the breast and negative nodes) at definitive surgery is unclear. Previous trials showed promising antitumor activity and an acceptable safety profile associated with pembrolizumab in patients with early triple-negative breast cancer. The most trusted, influential source of new medical knowledge and clinical best practices in the world. Information and tools for librarians about site license offerings. Valuable tools for building a rewarding career in health care. 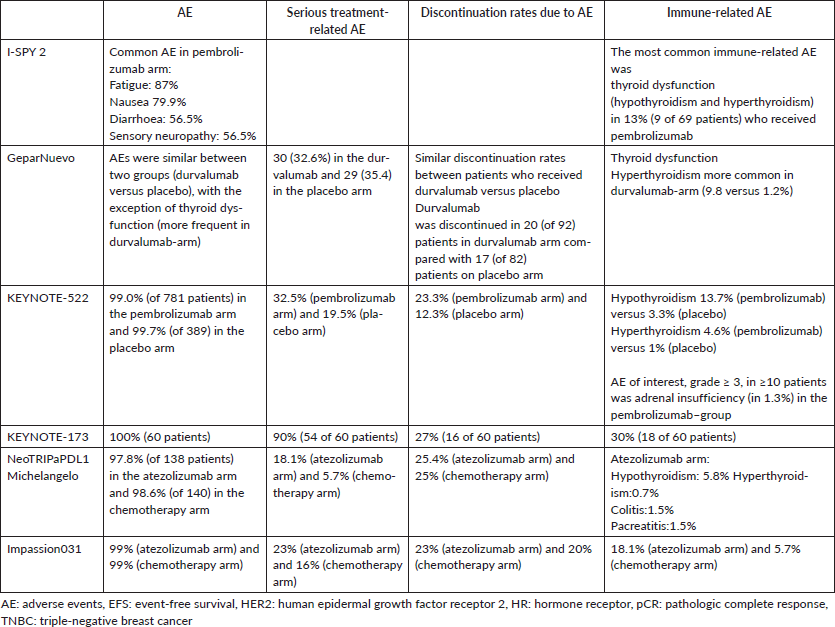
The authorized source of trusted medical research and education for the Chinese-language medical community. The most advanced way to teach, practice, and assess clinical reasoning skills. Information, resources, and support needed to approach rotations - and life as a resident. The most effective and engaging way for clinicians to learn, improve their practice, and prepare for board exams. NEW! Peer-reviewed journal featuring in-depth articles to accelerate the transformation of health care delivery.Ĭoncise summaries and expert physician commentary that busy clinicians need to enhance patient care. NEW! A digital journal for innovative original research and fresh, bold ideas in clinical trial design and clinical decision-making. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
